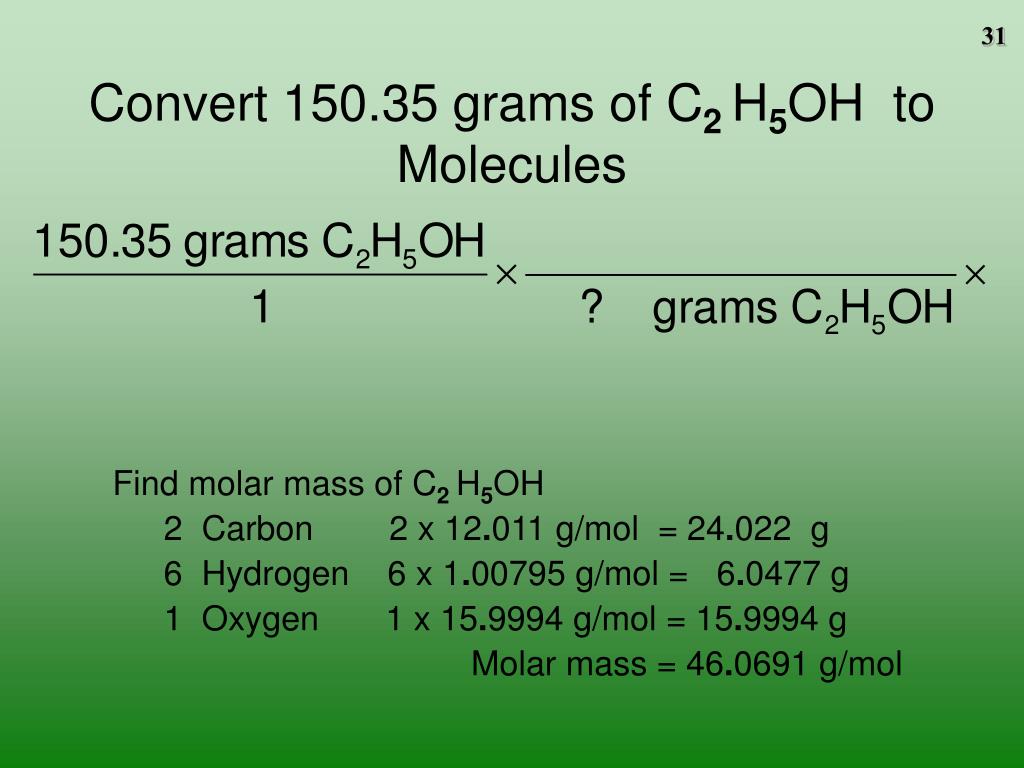
What is the formula for mass to moles? The formula for mass to moles conversion is: Moles = Mass (grams) / Molar mass (g/mol). So, 4g of H2 is approximately 2 moles.ĭo you use Avogadro’s number to convert grams to moles? No, you use Avogadro’s number to convert moles to molecules, atoms, or ions.ĭo you multiply or divide grams to moles? You divide grams by the molar mass to convert grams to moles.ĭo you use molar mass to convert grams to moles? Yes, molar mass is used to convert grams to moles. How many moles is 4 grams of H2? To find the number of moles, divide 4g by the molar mass of H2, which is approximately 2 g/mol. Stoichiometry involves using moles to quantify the relationships between substances in a chemical reaction. 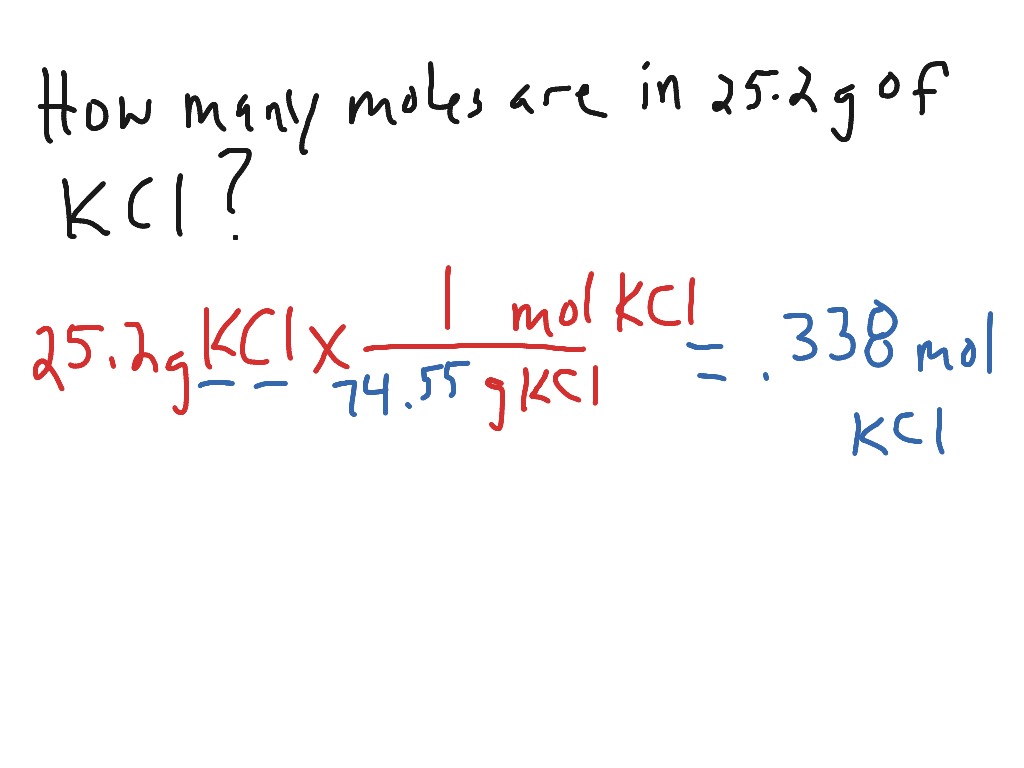
Is stoichiometry the same as moles? No, stoichiometry is not the same as moles. How to do stoichiometry for dummies? Stoichiometry for beginners involves understanding the balanced chemical equation, converting between moles and grams using molar mass, and using stoichiometric coefficients to relate quantities of reactants and products. Why do we convert grams to moles to begin a stoichiometry problem? We convert grams to moles in stoichiometry because it allows us to work with a consistent unit (moles) when using balanced chemical equations to relate reactants and products. How do you find grams in stoichiometry? In stoichiometry, you can find grams by converting moles to grams using the molar mass of the substance involved in the chemical reaction. It typically involves converting between moles of one substance to moles of another using balanced chemical equations. How do you convert stoichiometry? Stoichiometry involves the quantitative relationships between reactants and products in a chemical reaction. So, 50g of NaCl is approximately 0.86 moles. How many moles are in 50 grams of NaCl? To find the number of moles, divide 50g by the molar mass of NaCl, which is approximately 58.44 g/mol. How do you find moles? You can find moles by dividing the given mass (in grams) or the number of particles (molecules, atoms, ions) by the molar mass or Avogadro’s number, respectively. So, 20g of NaCl is approximately 0.34 moles. How many moles are in 20 grams of NaCl? To find the number of moles, divide 20g by the molar mass of NaCl, which is approximately 58.44 g/mol. The molar mass of a substance determines the number of grams in 1 mole. Is 1 mole equal to 40 grams? No, 1 mole is not equal to 40 grams. Is grams to moles one step? No, converting grams to moles typically involves two steps: calculating the molar mass and then dividing the given mass by the molar mass. The number of moles in 12 grams depends on the substance’s molar mass. Is 12 grams equal to 1 mole? No, 12 grams is not equal to 1 mole. 
This will give you the mass in grams.ĭo you calculate moles with grams? Yes, you can calculate moles from grams using the molar mass of the substance. How do you convert moles to grams for dummies? To convert moles to grams, multiply the number of moles by the molar mass of the substance. The choice depends on the given information and the specific stoichiometry problem. How do you find moles in stoichiometry? To find moles in stoichiometry, you need to convert either grams or molecules to moles using the substance’s molar mass or Avogadro’s number, depending on the given information and the specific problem.ĭo you have to convert to grams for moles? No, you can also convert from grams to moles or from molecules to moles. Use Avogadro’s number (6.022 x 10^23 molecules/mol) to convert moles to molecules.Convert grams to moles using the molar mass.How do you convert grams to molecules in stoichiometry? To convert grams to molecules in stoichiometry, follow these steps: So, 55g of NaCl is approximately 0.94 moles. How many moles is 55g of NaCl? To find the number of moles, divide 55g by the molar mass of NaCl, which is approximately 58.44 g/mol. The number of moles in 1 gram depends on the substance’s molar mass. Is 1 gram equal to 1 mole? No, 1 gram is not equal to 1 mole. How do I convert grams to moles? Divide the given mass in grams by the molar mass of the substance. Divide the given mass in grams by the molar mass to find the number of moles. The molar mass is the mass of one mole of a substance in grams. How do you convert grams to moles in stoichiometry? To convert grams to moles in stoichiometry, you need to use the molar mass of the substance you’re working with. This table provides a step-by-step guide for converting grams to moles in stoichiometry problems. Moles (mol) = Given Mass (g) / Molar Mass (g/mol) Look up the molar mass of the substance in g/mol.Įxpress the mass of the substance in grams (g). Identify the substance and its molar mass. Sure, here’s a table that summarizes the process of converting grams to moles in stoichiometry: Step Grams to Moles Calculator Mass (grams): Molar Mass (g/mol): Calculate Moles
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
